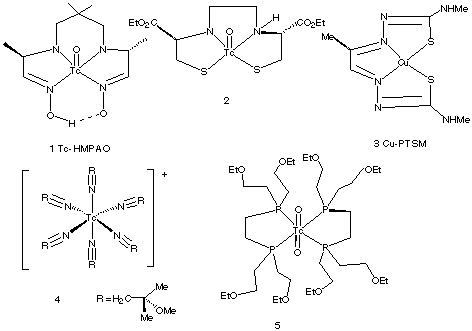
1.* (1997 F 4) Certain coordination compounds are used in clinical medicine as heart and brain imaging agents. A selection of these is shown below (from "Chemistry in Britain", October 1994, P. 820).
A. Indicate which compounds are chiral. (assume the two oxo groups in (5) are trans.). Write the compound numbers for any chiral compounds you find in Figure 2. B. Compound 2 enters the brain by diffusing through the brain blood barrier; this substance is trapped because it is acted on in the brain by an esterase. A stereoisomer of 2 is not trapped in the brain. Propose a plausible explanation of this fact. C. What is the oxidation state of copper in compound 3? Predict the coordination geometry, and d-electron configuration for 3. D. What is the oxidation state, d-electron configuration, and number of unpaired electrons in compound 4?
2.* (1997 2 2) A. Using any bidentate ("chelating") ligand of your choice, draw a structural formula for a first-row coordination complex which is both chrial and has three unpaired electrons. Also indicate the oxidation state, coordination number, and d electron count of the metal in your complex. Your structure should illustrate the chiral nature of this compound. B. Identify the 3d metal in the following diamagnetic complex anion: M(CN)64-. What is the oxidation state and d-electron count for your metal?
3.* (1997 2 3) Suppose you prepare a complex from propylenediamine, pn (H2NCH(CH3)CH2NH2) and rhodium: Rh(pn)3(ClO4) 3. A. What is the oxidation state of Rh and the d-electron count for this complex? B. Discuss the expected magnetic properties of this compound.
4.* (1996 F 1) What's the maximum number of unpaired electrons any transition metal complex can have? A. Illustrate this for a metal with oxidation state 2+. B. What other condition must be satisfied?
5.* (1996 F 2) Platinum II typically forms diamagnetic, square-planar complexes. Attempts to make octahedral Pt(II) complexes, even with a rigid multicyclic ligand which would force six nitrogens to bind in an octahedral fashion to Pt(II) have not succeeded even though such "sepulchrate" complexes of Co(III) are well known and are very stable. Use ligand field diagrams to explain the reluctance of platinum (II) to form octahedral complexes.
6.* (1996 F 11) A. If you want a transition metal compound to be a good oxidizing agent, should it have a lot of d electrons?
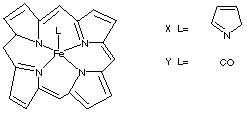
7.* (1996 F 13) Below are structural formulas for two iron porphyrins. The pyridine complex, X, is paramagnetic whereas the carbonyl derivative is diamagnetic. The pyridine complex strongly binds a second pyridine forming a diamagnetic bis-pyridine complex; however, the carbonyl complex, Y, only weakly binds a second carbonyl.
A. Offer two reasons for this difference. B. How many unpaired electrons are in the mono pyridine complex?
8.* (1996 2 1) Consider the following compounds; indicate the number of unpaired electrons in each. A. [Pd(NH3)4]2+ C. [Co(CN)5]3-
9.* (1996 2 3) Using only ethylene diamine (en = H2NCH2CH2NH2) and bromide anions as ligands construct a cationic octahedral complex of cobalt (III); your complex cation should have charge +1 and it should be chiral. Draw a three dimensional structure for this coordination complex below on the left. Then draw the structure of a diastereoisomer on the right.
10.* (1995 F 1) E. Give an example of a ligand which has neither p electrons nor non-bonded electron pairs. F. Give an example of a metal complex made only from an anionic aromatic ligand, show the structure, indicate the oxidation state of the metal and the d-electron count. H. Draw a stereochemical structure for a coordination complex which is chiral and has three unpaired electrons. I. Identify the 4d metal in the following diamagnetic complex: M(CO)6. Give its d-electron configuration.
11.* (1995 2 2) A. Draw all possible stereoisomers of [CoCl2(en)(NH3)2]+. Which are chiral? (en = H2NCH2CH2NH2) C. How many unpaired electrons are in the following complexes (assuming all of them have strong-field ligands?): Cu(NH3)42+, CrCl3(THF)3, K3Mn(CN)6.
12.* (1994 F 3C) Name a transition metal cation having 5 unpaired electrons.
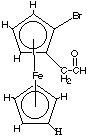
13.* (1994 F 7) A novel organometallic anion [Cu(CF3)4]- has recently been prepared. A. What is the shape of this anion? Explain your reasoning. Do you expect this substance to be diamagnetic? B. Calculate the oxidation state, the d electron configuration and the number of unpaired electrons in [N(Et)4]4[V(CN)6] (where Et = C2H5). C. Consider the following ferrocene derivative… What sort of stereoisomers would this substance have? How many stereoisomers would this substance have? How many different 1H NMR signals do you expect? Identify each by marking on the structure.
14.* (1994 2 1) For each of the following complexes: (a) calculate the formal oxidation state of the central metal, (b) give the d electron configuration, (c) deduce the coordination geometry, (d) predict the number of unpaired electrons and (e) give the number and types of steereoisomers, if any (indicate whether these stereoisomers are chiral). I. CoCl42- II. Fe(CN)63- III. Cr(acac)3 IV. Mo(CO)6
15.* (1994 2 5C) A. Describe how to resolve a racemic mixture of Ru(bipy)32+ into its two enantiomers, and how to make sure that your separation procedure has worked.
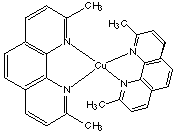
16.* (1993 F 1G) Name a complex cation with 3 unpaired electrons, a charge of 3+, and chirality at the metal center.
17.* (1993 F 5) 2,9-dimethyl orthophenanthroline is a reagent used for the selective binding of copper in one of its oxidation states. Which oxidation state of copper does this reagent favor? A copper complex of this ligand is shown below but the charge (and thus oxidation state) is not specified. Discuss this situation in the context of electronic structure and preferred coordination geometry.
18.* (1993 2 1) For each of the following complexes indicate the oxidation state of the central metal, the d electron configuration and the number of unpaired electrons, and whether the structure is chiral or achiral. A. Fe(CN)63- B. Ru(bipy)33+ C. Pt(CN)42- D. p-C6H5Mn(CO)3 E. Ni(PPh3)2(CO)2 F. Cr(acac)3
19.* (1993 2 3A) Which of the following is rouge and which is colorless? TiO2 and Fe2O3
20.* (1992 F 25) Consider the diamagnetic complex [M(en)3(ClO4) 3] where M is a 3d metal. A. What is M? B. What is the oxidation state of M? C. How many d electrons? D. Will this complex be a strong oxidizing agent? E. Sketch a picture of all stereoisomers of this complex. F. What features would the 13C NMR spectrum exhibit?
21.* (1992 2 4) Consider two complexes A and B. One is Ni(NH3) 2Br2; the other is Pd(OH2) 2Br2. A is paramagnetic and B is diamagnetic. A. What is the oxidation state of each of the metals? B. How many d electrons does each metal have? C. What is the coordination number of each complex? D. What is the geometry of each complex? E. Which is which? F. Identify the stereoisomers (if any) of each complex.
22.* (1991 2 2) Consider (Cp)M(CO)x. What is M for X=1, X=2, and X=3?
23.* (1991 2 3) For each of the following complexes, give the oxidation state, number of d electrons, coordination number, number of possible isomers, and state whether the complex is chiral: A. Co(en)33+ B. Pt(NH3)2(Br)2 C. Ni(CO)4 D. Re(CH3)(CO)5
24.* (1991 2 4) Using any ligand and 3d metal write the formula of a chiral complex with 3 unpaired electrons. Indicate overall charge, oxidation state, and dn.
25.* (1991 2 5) A. Sketch the two enantiomers of Ru(bipy)32+. B. Sketch the bipy ligand. C. How many signals does the proton-decoupled 13C NMR of Ru(bipy)32+ have? D. How many magnetically inequivalent types of H atoms does Ru(bipy)32+ have? E. Is this molecule chiral?
26.* (1990 F 12) Consider a pair of isomeric cationic complexes having the molecular formula [Co(en) 2Br2]ClO 4. One is optically active but the other is not.
|