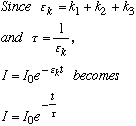Phototherapy 1.* (1997 F 12) Ferrochelatase is an enzyme which inserts iron into protoporphrrin IX; the iron complex is then taken up by hemoglobin. Some people are deficient in this enzyme and have some iron free protoporphyrin IX. Such individuals develop dark lesions on their skin when they are in the sun. Explain the underlying cause of this disease in terms a Chem 31 student would understand. Do you expect this disease to be congenital? Porphyrins are excellent photosensitizers, exciting oxygen from triplet to singlet O2, using light energy (hv). Singlet oxygen is very reactive, and will oxidize funcionalities in the person's cells, etc, leading to the skin lesions. Paramagnetic metals in porphyrins quench singlet oxygen. So, the lack of ferrochelatase, which inserts iron into protoporphyrin IX, is a problem because, without the iron, the singlet oxygen is not quenched. This enzymatic defect is congenital. Enzymes are made by coding in the DNA, so this defect is inherited. 2.* (1995 F 1) A. Discuss the ground state and lowest excited state of O2.
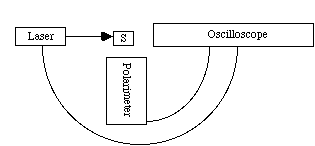
B. Why are metal-free porphyrins useful in photodynamic therapy while metal-containing porphyrins are not? Porphyrins absorb visible light, and are thus excited into a singlet state: The excited porphyrin then reacts with triplet oxygen to form singlet oxygen: O2 (singlet) is the active material for destroying cell walls, and is vital to photodynamic therapy. With a metal present, there is a rapid quenching of O2 (singlet) and therefore no photodynamic therapy. 3.* (1990 F 4) A chemist proposed a catalyst to help prepare singlet oxygen from oxygen. If you were his research director what advice would you offer? This reaction is thermodynamically unfavorable (a catalyst only brings you to equilibrium distribution faster). Therefore, there’s no point in catalyzing. Tell the chemist to go back to grad school or something. Partial credit was given if you showed how a photosensitizer such as Ru(bipy)32+ promotes this process without being used up. The fact that you need a constant input of light means that this process is not technically catalytic. 4.* (1990 F 6) How do you determine the unquenched radiative lifetime T0 of Ru(bipy)3Cl2 dissolved in water? •First we must excite to T1. •T0 implies that no quenchers are present, so the solution must be purged with an inert gas (N2, He, Ne) to remove dissolved oxygen. Also use a solution saturated in O2 and saturated with air. •Use the experimental setup we used in lab:
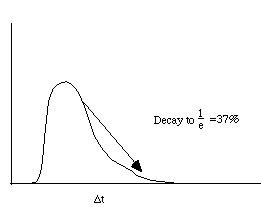
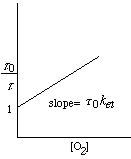
•Measure the time it takes to decay to • •Use Stern-Valmer plot to find •
|